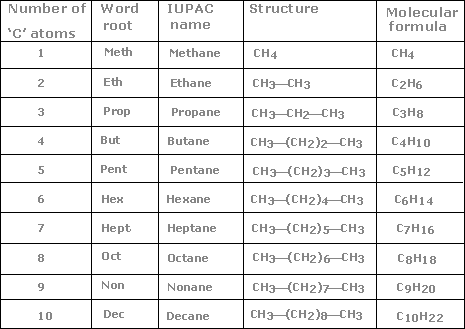
Numbering of the various substituents and bonds with their locants. Locants are the numbers on the carbons to which the substituent is directly attached.ī) Has the lowest-numbered locants for multiple bonds (The locant of a multiple bond is the number of the adjacent carbon with a lower number).Ĭ) Has the lowest-numbered locants for prefixes. This is done by first numbering the chain in both directions (left to right and right to left), and then choosing the numbering which follows these rules, in order of precedence.Ī) Has the lowest-numbered locant (or locants) for the suffix functional group. When both side chains and secondary functional groups are present, they should be written mixed together in one group rather than in two separate groups. The "di" is not considered in either case). For example, ethyl comes before dihydroxy or dimethyl, as the "e" in "ethyl" precedes the "h" in "dihydroxy" and the "m" in "dimethyl" alphabetically. are not taken into consideration for grouping alphabetically. Identification of the remaining functional groups, if any, and naming them by their ionic prefixes (such as hydroxy for -OH, oxy for =O, oxyalkane for O-R, etc.).ĭifferent side-chains and functional groups will be grouped together in alphabetical order. Side chains are the carbon chains that are not in the parent chain, but are branched off from it. Identification of the parent functional group, if any, with the highest order of precedence. If more than one functional group is present, the one with highest precedence should be used.Ī) It should have the maximum number of multiple bonds.ī) It should have the maximum number of single bonds. 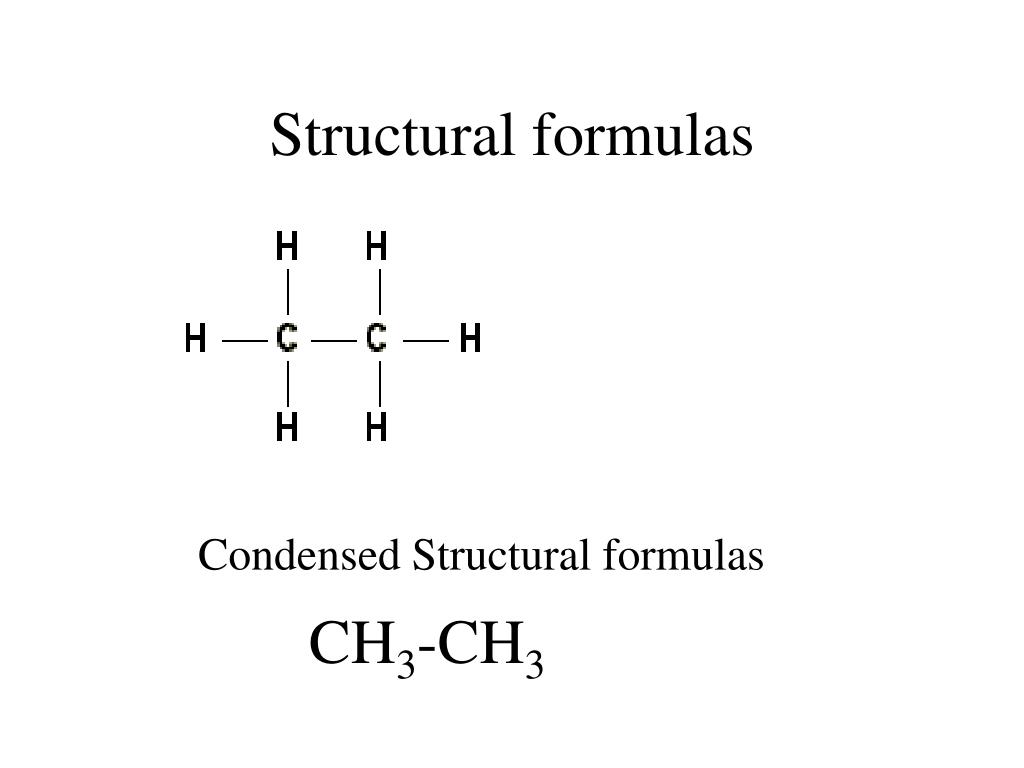
By suffix, it is meant that the parent functional group should have a suffix, unlike halogen substituents. It should have the maximum number of substituents of the suffix functional group. This chain must obey the following rules, in order of precedence: Identification of the parent hydrocarbon chain. The steps for naming an organic compound are as follows: Which, if you ask me, sounds ugly, so I’m never going to repeat it again.In organic chemistry, a number of prefixes, suffixes and infixes are used to describe the type and position of functional groups in the compound. The more “correct” definition of HDI is the number of H 2 equivalents required to saturate molecule to have only open-chain single bonds structure. Recall that double and triple bonds have π-bonds. Basically, each degree of unsaturation or HDI unit corresponds to either a π-bond or a cyclic motif in the molecule. HDI is a useful tool to estimate the structural motifs in the molecule. Anyways, back to HDI and why it’s so cool. And frankly, I’m a little confused why it’s virtually never taught here. While HDI is usually not introduced till we look into spectroscopy, I believe, it’s a very potent tool to know early in the semester. The exact term your instructor will use will depend on their personal preferences or the book choice. Hydrogen Deficiency Index (HDI)īoth of those terms refer to the same thing. You’ll see examples of constitutional isomers many times in your course, so it’s very important to be able to spot those. The important thing about the constitutional isomers, is to realize that those are different molecules and, therefore, have different properties. However, the connections between those atoms, or in other words, the constitution of the molecule, is different. They both have exactly the same atoms in the same ratios in the molecule. The two molecules above, ethanol (ethyl alcohol) and dimethyl ether, are example of constitutional isomers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
